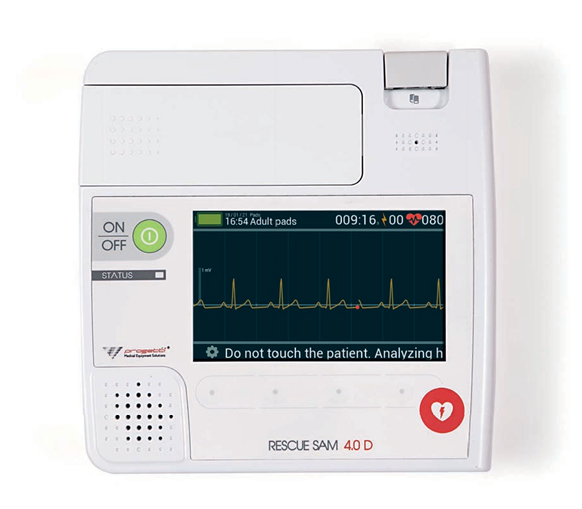
Descriptions:
Rescue SAM 4.0’s smart connection system recognises the plates and sets the shock to the most appropriate protocol for the adult or paediatric patient .The special connector has been made to ensure the best construction standards.
Rescue Sam 4.0D was designed to also be used in the most extreme emergencies. It was therefore subjected to countless tests by accredited, avant-garde laboratories, who worked to ensure that our devices are durable, sustainable and safe and that they can withstand shocks, impacts and the entry of dust and liquids (IP55).
TECHNICAL SPECIFICATIONS:
DEFIBRILLATOR
How to use: semi-automatic, for adult and paediatric use.
Waveform: Biphasic Truncated Exponential (BTE) controlled by impedance measurement.
Energy:
adults: ±xed at 200J – paediatric: less than 50J Charging time from discharge warning: <8 seconds to 200J with new battery; <5 seconds at 50J on paediatric patients.
Pacemaker detection: detection and removal of the artefact created by the pacemaker
DISPLAY
Type: 7’’ colour LCD, resolution 800×480 pixels Brightness control: 10 settable levels
ECG
Band: ECG displayed: 0.25 – 160 Hz
CMRR: 120 dB
Input impedance: ›500MΩ
Amplitude: 25 mm/mV
Speed: 37.5 mm/s
Filters: ±xed ±lters for removing supply disturbances, baseline wandering and signal conditioning
HR frequency range: 20 – 300 bpm
PHYSICAL & ENVIRONMENTAL
Dimensions: 250 x 260 x 80 mm
Weight: 2.2 kg including battery and standard accessories
Protection class: IP 55
Operating temperature: -20°- 60 °C
BATTERY
Characteristics: 12V DC, 4200mAh non-chargeable long-lasting lithium-manganese battery, internal memory.
Capacity: up to 12 hours in continuous operation or up to 200 shocks at maximum energy 200 J
AUTOMATIC SELF-TEST
Battery insertion test, Daily, Weekly, Monthly Tests, Battery check
DATA RECORDING AND TRANSFER
Internal memory: up to 16 hours recording ECG tracing and events
Environmental recording: ›2 hours (optional)
CONNECTIVITY
USB: standard, micro-B
STANDARDS AND SAFETY
IEC standard: EN 60601-1; EN 60601-1-1;
EN 60601-1-2; EN 60601-2-4
EC mark: EEC 93/42, medical device
Product CatalogueRelated products
-
Blood Fluid Warmer Astramed UK
₨175,000.00 -
Ambu® PEEP Valves – Disposable and Reusable
Call for Price -
Fetal monitors – FM20
Call for Price